What's Happening
March 02, 2026
Meridian Introduces Oscillometry Testing at Cedar County Memorial Hospital
Meridian, a Lincare affiliate and the nation’s leading provider of pulmonary rehabilitation and diagnostic services, is proud to announce the addition of airway oscillometry testing at its centers. Meridian recently expanded its clinical services at, Cedar County Memorial Hospital in El Dorado Springs, Missouri marking its 40th facility to provide oscillometry testing and enhancing rural healthcare.
READ MOREJanuary 29, 2026
Medicare Announces Updates to Competitive Bidding Program
On November 28, 2025, Medicare released its Final Rule for the Competitive Bidding Program, outlining changes for the next round of bidding. The update primarily focuses on newer product categories, while Lincare’s core products, such as oxygen therapy and CPAP equipment, will not be included.
READ MOREJanuary 22, 2026
Community’s Choice: Lincare Lynchburg Earns Gold Honors for 18th Consecutive Year
Lincare proudly celebrates its Lynchburg VA center being named the Best Medical Equipment & Supply provider for Lynchburg City and surrounding counties by Living in Lynchburg magazine. This marks the 18th consecutive year our team has earned this prestigious honor.
READ MOREJanuary 15, 2026
Linde & Lincare Celebrate Giving Tuesday with Global Impact
Lincare’s parent company, Linde, continues to make a difference in communities worldwide through its year-round Global Giving program. This year, in honor of Giving Tuesday, a global generosity movement launched in 2012 to inspire people to do good, Linde invited employees to share nonprofit organizations that hold special meaning to them.
READ MOREDecember 23, 2025
Back-to-Back Champions: Hays Lincare Named Best of the Best in Health Care
We are thrilled to announce that our Hays, KS Lincare Center has once again been recognized as the “Best of the Best” in the Health Care category by the Hays Daily News. This marks the second consecutive year our team has earned this prestigious honor, a clear reflection of their dedication to excellence and the trust they’ve built within the community.
READ MOREDecember 17, 2025
Making Spirits Bright: Lincare HQ Comes Together for Silver Bells & Angel Tree
This holiday season, once again we have come together to sponsor individuals through The Salvation Army’s Silver Bells and Angel Tree programs, bringing joy to seniors and children who might otherwise go without during the holidays.
READ MORESeptember 26, 2025
Lincare HQ Hosts Second Annual Art Auction to Support Hands Across the Bay
Lincare’s Corporate Finance team recently hosted its second annual art auction fundraiser, expanding the event to include contributions from across HQ. This year, employees were invited to create and donate original artwork in a variety of forms—from paintings and photography to handcrafted items. The pieces were beautifully displayed at the café entrance, and bidding took place through a custom online form.
READ MORESeptember 09, 2025
Lincare Supports Malnutrition Awareness Week 2025: September 8–12
Malnutrition Awareness Week™ 2025, taking place from September 8–12, is a vital initiative led by the American Society for Parenteral and Enteral Nutrition (ASPEN). This annual campaign, established in 2012, brings together healthcare professionals, caregivers, and communities to spotlight the serious impact of malnutrition and promote better care for those affected.1
READ MOREJuly 01, 2025
Local ALS Walk Celebrates Community Unity and Support
Centers from the southeast Pennsylvania region came together to participate in the annual ALS United Mid- Atlantic Walk, demonstrating their commitment to raising awareness and support for those affected by Amyotrophic Lateral Sclerosis (ALS). This year’s event highlights the powerful role of local and corporate supporters, with Lincare receiving recognition of their contributions.
READ MOREJune 11, 2025
Local Lincare Centers Visit ALS Clinic and Research Lab
Staff from the Southeast Pennsylvania Lincare Centers received a unique opportunity to deepen their understanding of Amyotrophic Lateral Sclerosis (ALS) through an exclusive tour of the ALS Clinic and Research Center at the Penn State College of Medicine.
READ MOREJune 02, 2025
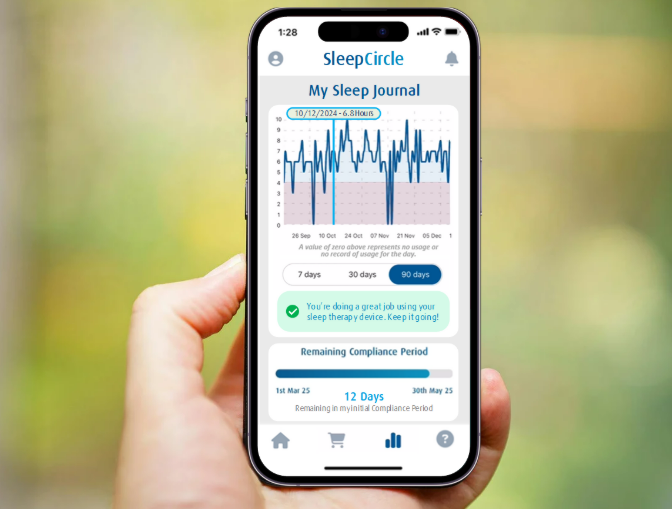
Announcing the Launch of SleepCircle: Lincare's New Mobile App for Sleep Therapy Patients
Lincare is excited to announce the launch of SleepCircle®, our innovative mobile application designed to enhance the experience and support of our sleep therapy patients. This easy-to-use app delivers six core functions specifically tailored to meet the unique needs of our patients, ensuring they receive the highest standard of care and support.
READ MOREMay 28, 2025
Lincare Heroes in the Sky: A Life-Saving Flight to Remember
This week, the Lincare family celebrates an extraordinary act of heroism by two of its own: Bobby and Gabby. These remarkable individuals, who recently completed corporate training, demonstrated their skills and compassion by saving a life mid-flight.
READ MOREMay 15, 2025
Lincare Embraces Global Safety Commitment Week
During the week of May 12–16, Lincare proudly joins our parent company, Linde, in celebrating Global Safety Commitment Week. This dedicated time allows us to renew our focus on safety, even as we continue to make significant strides in enhancing our safety performance each year.
READ MOREMay 01, 2025
Lincare Enteral Team Attends ASPEN25 Conference
Last month, members of the Lincare Enteral team had the privilege of attending the American Society for Parenteral and Enteral Nutrition (ASPEN) annual conference. This prestigious event brought together leading clinical nutrition professionals from across the country, offering a platform for exchanging insightful ideas and best practices.
READ MOREApril 16, 2025
Lincare's Commitment to Excellence: Reaccreditation by CHAP
Every three years, Lincare must renew its accreditation with CHAP (Community Health Accreditation Partner), an independent, nonprofit accrediting body for home and community-based healthcare organizations. We are proud to announce that Lincare has been reaccredited by CHAP, effective December 15, 2024, for our Home Medical Equipment and Pharmacy services, including our wholly owned subsidiaries.
READ MOREApril 09, 2025
Celebrating Angie: A Pillar of Community Advocacy and Excellence
On Saturday, March 8th, Angie, a dedicated Health Care Specialist at Lincare in Marlton, NJ, was honored at the Salem County Commission of Women's 2025 Women of Achievement Recognition Breakfast. This prestigious event celebrated individuals who have made significant contributions to their communities. The theme for the event was "Power and Presence," a perfect encapsulation of Angie's impact in Marlton.
READ MOREMarch 06, 2025
Jeff’s Journey to Health: A Successful Pulmonary Rehabilitation from Jenkins County Medical Center
Two years ago, Jeff took a courageous step that would forever change his life. Battling the debilitating effects of long COVID, COPD, and lungs that were 40% scarred, Jeff found even the simplest tasks nearly impossible. Walking 20 to 30 yards required the constant support of a portable oxygen tank, and his once-active life was profoundly altered.
READ MOREFebruary 26, 2025
Introducing REACH: Lincare's Employee Resource Group
Lincare is excited about the establishment of a Employee Resource Group (ERG) chapter: REACH (Resources for Employees of African Culture & Heritage). This initiative marks a significant step in fostering an inclusive and supportive environment for all employees, particularly those of African American heritage. Our mission is to create a workplace where diversity is celebrated, and every individual feels valued and empowered.
READ MOREFebruary 19, 2025
Hays, KS Lincare Takes the Crown in "Best of The Best" local Contest
At Lincare, we are thrilled to announce that our Hays, KS office has won in the "Best of The Best" contest.
READ MOREFebruary 13, 2025
Lincare's REACH ERG Group Attends MLK Dream Big Banquet
St. Petersburg, FL – Several members of Lincare's Employee Resource Group (ERG), REACH (Resources for Employees of African Culture & Heritage), proudly attended the prestigious MLK Dream Big Banquet on Friday, January 17, 2025.
READ MOREFebruary 05, 2025
Lincare Chatsworth Team: Rising to the Challenge During Wildfires
In recent weeks, the Lincare team in Chatsworth, California, has exemplified resilience, teamwork, and compassion in the face of adversity. As wildfires ravaged the Chatsworth/Los Angeles area, the team not only safeguarded their own members but also provided crucial support to patients in need.
READ MOREFebruary 04, 2025
Patient Success Story: A Journey to Restful Nights
In the world of sleep medicine, few moments are as heartwarming as witnessing a patient's transformation. A recent visit from a patient to our Marlton, NJ center was one such moment.
READ MOREJanuary 29, 2025
Vanessa Bull's Success Story: Breathing New Life with Dual Therapy
Living with Chronic Obstructive Pulmonary Disease (COPD), Chronic Respiratory Failure (CRF) and bronchiectasis can be a daunting challenge, but for Vanessa Bull, an innovative dual therapy approach has transformed her life.
READ MOREJanuary 22, 2025
Lincare Centers in Lynchburg: Celebrating Excellence with a Gold Award
Lincare centers in Lynchburg, VA, have proudly been recognized with the gold award in the Lynchburg Living – 2024 Best of Readers' Choice Award for Best Medical Equipment & Supply Company. This recognition, voted on by the community, highlights the Lincare centers’ dedication to quality healthcare.
READ MOREJanuary 07, 2025
Blood Drives and Community Impact: Lincare's Commitment to Health and Wellness
We recently marked the successful conclusion of our final blood drive of the year at our headquarters located in Clearwater, Florida. This event was a meaningful collaboration with OneBlood, a not-for-profit organization dedicated to enhancing health and well-being through blood donation services.
READ MOREDecember 20, 2024
Lincare Spreads Holiday Cheer with Successful Gift Drive in Partnership with The Salvation Army
Lincare headquarters has once again spread holiday joy through a heartfelt gift drive, highlighting the generosity and compassion of its employees.
READ MOREDecember 17, 2024
Lincare HQ Unites for Thanksgiving Food Drive
As the spirit of Thanksgiving embraces the nation, Lincare Headquarters has taken a heartwarming initiative to give back to the community. In collaboration with the Salvation Army, Lincare organized a general drop-off food drive, demonstrating their commitment to aiding those in need during the holiday season.
READ MORENovember 25, 2024
Lincare Enteral Division Shines at Upstate University Hospital DME Fair
The Upstate University Hospital recently hosted its annual Durable Medical Equipment (DME) Fair, a key event for healthcare providers and suppliers to showcase their innovations and services.
READ MORENovember 15, 2024
Understanding Our Social Media Engagement
Our team is dedicated to fostering a supportive and engaging online community where you feel heard and valued. We understand the importance of building trust and genuine connections, which is why we prioritize human interaction over automated responses. Our aim is to create a welcoming space where your questions, comments, and feedback are met with care and consideration.
READ MORENovember 13, 2024
Lincare Responds to the Devastation Caused by Hurricanes Helene and Milton
In the aftermath of Hurricanes Helene and Milton, which caused significant destruction across several states, including Tennessee, North Carolina, Georgia, and Florida, we, as a leading provider of respiratory therapy services, have taken action to assist the impacted communities by offering essential healthcare services and resources.
READ MORENovember 08, 2024
Lincare Long-Term Care Team's Response to Hurricane Devastation
In the wake of recent devastating hurricanes that swept through various regions, Lincare's Long-Term Care division demonstrated exceptional commitment and resilience in supporting affected facilities. Understanding the critical needs of nursing homes and their residents, the division sprang into action, ensuring that essential supplies, particularly oxygen tanks, reached those in dire need.
READ MOREOctober 31, 2024
Lincare Launches Hurricane Helene Support Drive
Supporting Hurricane Helene relief through employee support drive.
READ MOREOctober 29, 2024
Celebrating Hispanic Heritage Month: A New Initiative by the REACH ERG Group
This Hispanic Heritage Month, Lincare’s newly formed REACH Employee Resource Group (ERG) took the opportunity to honor the contributions of the Hispanic community and promote cultural awareness within the Company. Through a series of engaging activities, the group facilitated a deeper understanding of Hispanic heritage among employees.
READ MOREOctober 25, 2024
Celebrating Excellence: Honoring Our Respiratory Professionals During Respiratory Care Week
Respiratory Care Week is a significant event that shines a spotlight on the vital role respiratory therapists play in healthcare. Celebrated annually, it offers an opportunity to recognize these dedicated professionals and their contributions to improving patients' respiratory health.
READ MORESeptember 30, 2024
Lincare Employees Unite for 9/11 Day of Remembrance Meal Pack Project
Check out Lincare employees as they volunteer for 9/11 Day, honoring those affected by the tragedy through acts of kindness and community service.
READ MORESeptember 04, 2024
William Newton Memorial Hospital Celebrates Another Successful Meridian Pulmonary Rehab Graduate
Celebrate the Meridian Pulmonary Rehab Program at William Newtown Memorial Hospital. Get personalized care, expert guidance, and support for a healthier, more active life through pulmonary rehabilitation services.
READ MOREJune 11, 2024
Jeff Barnhard, CEO of Lincare, Named Chairman of the Council for Quality Respiratory Care
The Council for Quality Respiratory Care (CQRC) – a coalition of the nation’s leading home respiratory therapy providers and manufacturing companies – recently named Jeff Barnhard, chief executive officer of Lincare, as Chairman of the Board.
READ MOREMay 30, 2024
Lincare Recognized for Outstanding Safety Achievements in 2023
We are thrilled to announce that Lincare, a part of Linde, has been honored with the prestigious 2023 Safety Awards for its exceptional progress towards achieving "Zero Today" in safety.
READ MOREMay 08, 2024
Lincare.com Launches New Chat Feature to Enhance Patient Experience
We are excited to announce the launch of our new chat feature, which allows patients to communicate directly with our team of professionals. This new feature is now available for use, and we hope it will enhance the patients' experience with Lincare.
READ MOREMay 03, 2024
Blood Drives at Lincare Touch 123 Lives
At Lincare, we strongly believe in giving back to our community and making a positive impact wherever possible. That’s why we partnered with OneBlood on Wednesday, April 25th, for a blood drive at our headquarters in Clearwater, Florida.
READ MOREApril 30, 2024
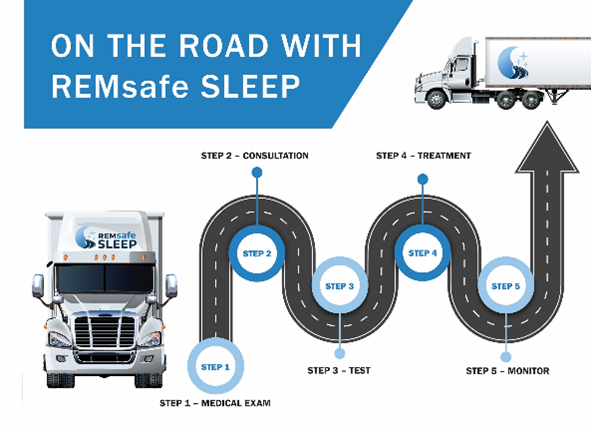
Lincare's REMsafe SLEEP® Secures New Business with TCW
REMsafe SLEEP®, one of our family of businesses and a leading provider of sleep apnea solutions, has announced a new business relationship with TCW, a leading warehousing and distribution company. This relationship will provide offer TCW's fleet of 500 drivers with state-of-the-art sleep apnea treatment.
READ MOREApril 23, 2024
Lincare Supports the SOARS Act to Ensure Access to Respiratory Care Services
Lincare is a leading provider of respiratory care services and is proud to support the Stabilizing Oxygen Access for Rural Seniors (SOARS) Act. This bill, which has bipartisan support, aims to improve access to home oxygen services for vulnerable populations, especially those living in rural areas.
READ MOREApril 08, 2024
Lincare Volunteers Participate in Community Engagement Beach Cleanup Project
A team from Lincare headquarters recently participated in a community engagement activity aimed at promoting environmental conservation and community support. The Pier 60 Sugar Sand Festival Cleanup took place on Sunday, April 7, at Clearwater Beach, FL.
READ MOREMarch 19, 2024
Lincare Revolutionizes Patient Care With the Launch of Its New Patient Portal
Lincare, a leading provider of home respiratory care, is proud to announce the launch of its innovative Patient Portal, a cutting-edge platform designed to enhance the patient's experience and improve healthcare communication.
READ MOREFebruary 28, 2024
Notice of Privacy Incident Related to Philips Respironics
A message for patients using Respironics sleep and respiratory devices concerning a recent Respironics third-party vendor privacy incident that may have impacted certain patient personal information.
READ MOREDecember 27, 2023
Meridian’s Successful Pulmonary Rehabilitation Services at William Newton Memorial Hospital
Discover the incredible success story of Meridian’s Pulmonary Rehab program at William Newton Memorial Hospital. See how they’ve revolutionized the field with their cutting-edge techniques and exceptional patient care.
READ MOREJuly 18, 2023
Jeff Barnhard Appointed Chief Executive Officer of Lincare
Our parent company Linde has announced Mr. Jeff Barnhard as the new Chief Executive Officer of Lincare Holdings Inc. (Lincare), Linde’s respiratory supply business in the US, based in Clearwater, Florida.
READ MOREApril 18, 2023
Lincare’s Preparation for Certain PHE Waivers Lifts
Discover how to navigate the upcoming PHE Waiver terminations together.
READ MOREMarch 01, 2023
Enteral Formula Shipment Delays
Over the past year, Lincare has been managing manufacturer-driven enteral formula shortages.
READ MORESeptember 27, 2022
Lincare Releases Statement About Hurricane Ian
This is a safety message from Lincare regarding Hurricane Ian.
October 01, 2020
A Prescription for Change
How Lincare is leveraging the power of digital and data to upscale its homecare services.
READ MOREAugust 10, 2022
Abbott - Urgent Expanded Voluntary Product Recall
Lyons Magnus, a third-party manufacturer for Abbott, is expanding its voluntary recall initiated on July 28, 2022 to include Ensure Harvest, PediaSure Harvest and additional lots of Club Pack Glucerna Original Vanilla, Chocolate, and Strawberry in 8 fluid oz cartons. No other Abbott products are impacted by this voluntary recall.
READ MOREFebruary 21, 2022
Abbott - Urgent Voluntary Product Recall
An important message to Lincare patients about the Abbott urgent voluntary product recall powder formula.
READ MOREAugust 16, 2022
Kate Farms - Urgent Expanded Voluntary Product Recall
An important message to Lincare patients about the Kate Farms urgent voluntary product recall.
READ MOREMarch 19, 2020
Lincare Releases Statement About COVID-19 Commitment
COVID-19 Updates. Steps we are taking to protect you.
March 25, 2020
Lincare Statement Regarding COVID-19
Lincare, a subsidiary of Linde (NYSE:LIN; FWB:LIN), the leading provider of home respiratory care in the U.S., stands as a second line of defense in support of efforts to address the COVID-19 (coronavirus) public healthcare crisis. Throughout this pandemic, Lincare remains committed to safely and reliably provide critical in-home services.
READ MORELincare Releases Video Statement About COVID-19 Commitment
Lincare released a video that shows how our mission is more important than ever at this time of COVID-19. Our critical role helps to maximize hospital capacity while we care for patients in their homes. We stand with our fellow healthcare providers, dedicated to an excellent standard of care.
READ MORESeptember 08, 2022
Philips™ Respironics alerts customers of updated instructions and labeling of specific sleep therapy masks that contain magnetic headgear clips due to potential risk of serious injury
An important message to Lincare patients with Philips™ Respironics devices that contain magnetic headgear clips.
READ MORE